Zoom Details:
https://stanford.zoom.us/j/97187544962?pwd=blFuOE81OFVkQ29LbDB0dWZZbk5wdz09
Password: 496886
Zoom Details:
https://stanford.zoom.us/j/97187544962?pwd=blFuOE81OFVkQ29LbDB0dWZZbk5wdz09
Password: 496886


Our laboratory is focused on understanding the dynamics of organelle motility along the cellular cytoskeleton, driven by molecular motors: cytoplasmic dynein, kinesins, and myosins. The active transport of membrane-bound organelles is required for intracellular biosynthetic and endocytic trafficking in most eukaryotic cells. However, the highly polarized morphology of neurons, with axons that can extend over distances of up to one meter, make these cells uniquely dependent on motor-driven transport.

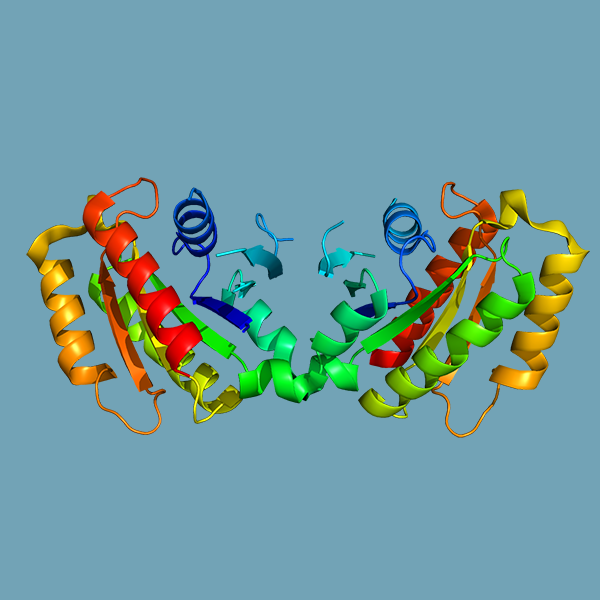
Mutations in Leucine-rich repeat kinase 2 (LRRK2) have been linked most infamously to Parkinson’s disease but also to a host of chronic inflammatory diseases, cancer, and susceptibility to infection; however, the underlying mechanisms through which LRRK2 mutations promote disease are unknown. We discovered that macrophages harboring the common human mutation Lrrk2G2019S are more prone to regulated cell death in response to both Mycobacterium tuberculosis (Mtb) infection and canonical inflammasome activation.

Mutations in leucine-rich repeat kinase 2 (LRRK2) are commonly implicated in the pathogenesis of both familial and sporadic Parkinson's disease. LRRK2 regulates critical cellular processes at membranous organelles and forms microtubule-based pathogenic filaments, yet the molecular basis underlying these biological roles of LRRK2 remains largely enigmatic.